Awards





















Timeline
Comparison
Market
Traditional bladder cancer diagnostics and treatment methods are expensive. Urinary bladder cancer has the highest per patient lifetime treatment costs of any cancer. The paper ”Economic Burden of Bladder Cancer Across the European Union” estimates that bladder cancer cost EU €4.9 billion in 2012. This amounted to 3 percent of all cancer costs.
It is estimated that upwards of $5.71 billion will be spent on bladder cancer in the US in the year 2020. The high cost shows the great need to improve the management of bladder cancer for the benefit of patients, healthcare systems and payers.
Today’s methods are both labor intensive and time consuming.
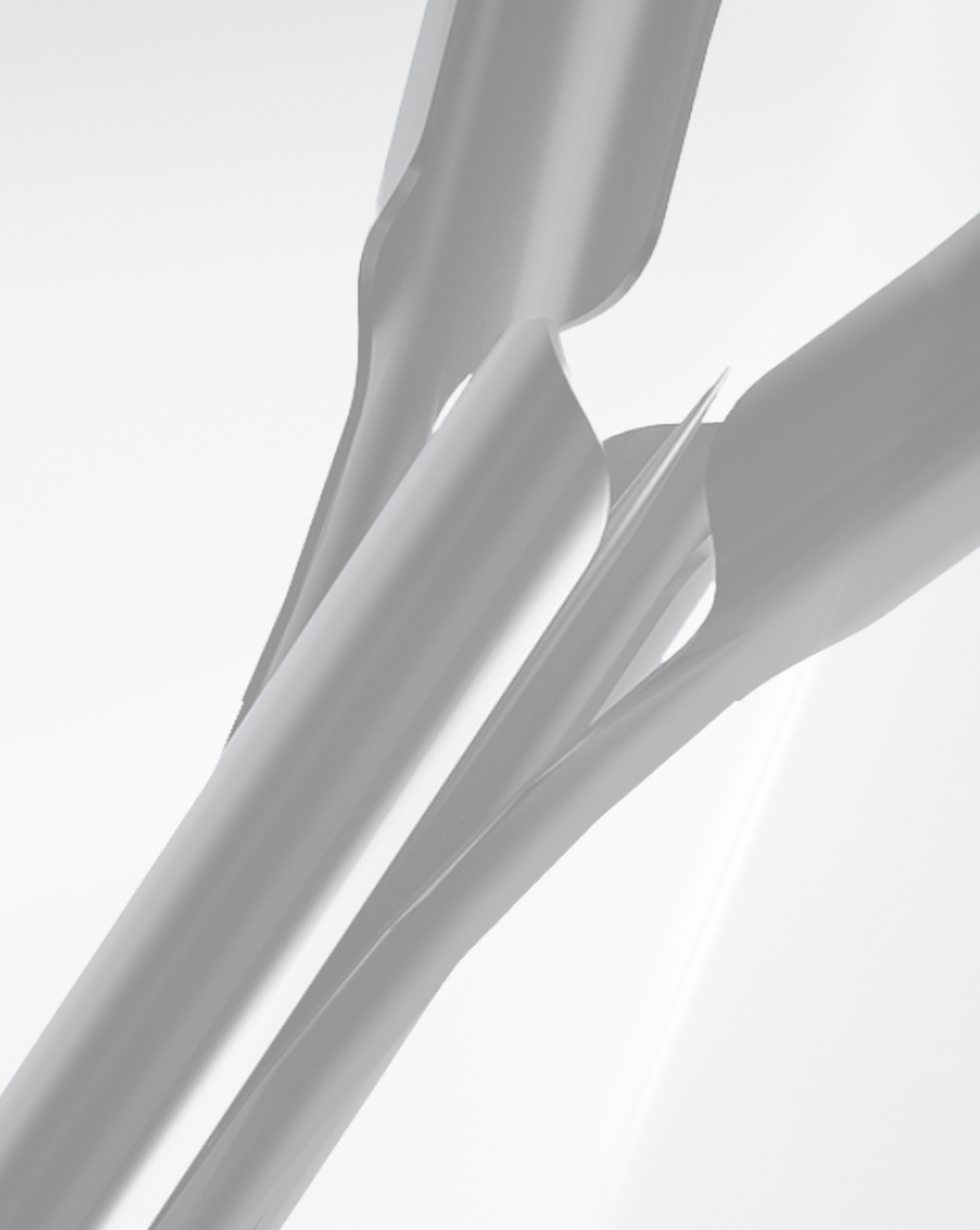
Multi4 cuts away many steps while adding value in the process.
Treatment is performed at the first visit in just 30 minutes in an outpatient setting.
There are many surgeries performed annually for bladder cancer, not least because the recurrence rate is so high. In the EU5 countries, 360,000 surgeries and in the United States, 315,000 are performed annually (transurethral resections of bladder cancer).
In addition, the world new Multi4-technology is scalable and can be used for cancer in other organs. The global market for endoscopes and associated equipment has an annual turnover of EUR 22,5 billion. Approximately 30% of this market. EUR 7 billion, consists of endoscopy accessories.
The market for diagnostic endoscopic biopsy instruments is expected to continue to grow from approximately EUR 1.5 billion in 2016 to approximately EUR 2.2 billion in 2022. Unfortunately, the number of cancer patients is increasing annually, new screening programs are introduced and the need for tissue sampling for diagnostics is increasing.
Multi4 collaborates with RISE – The research institute of Sweden.
RISE is a strong innovation partner who provides comprehensive support and a broad range of perspectives. RISE coordinates innovation research on Multi4 and assists with important regulatory expertise.
We work in parallel with many processes instead of serially to ensure a fast and high-quality journey towards launch so patients can get a treatment of their cancer without waiting times, in an easier way with less suffering , while healthcare and society save millions.
Don’t hesitate to contact us if you want to join our journey to fight cancer and reduce suffering for cancer patients around the world!
A broad and committed team
We are many who fight cancer together. We work with passion and we have a higher purpose.
In addition to a very driven management team that works operationally with a team of engineers, there are many people involved in the work every day. We work closely with two different patent office’s specializing in Med Tech to ensure a robust IP-portfolio.
Always available and so incredibly important are all colleagues in the healthcare sector, doctors, nurses, endoscopists and assistant nurses, but also directors, managers and professors who are engaged on demand from the start and help us move forward. You are all worth gold.
Multiple Prize Winner.
Proudly Funded by Contributions from Sweden & the European Union.